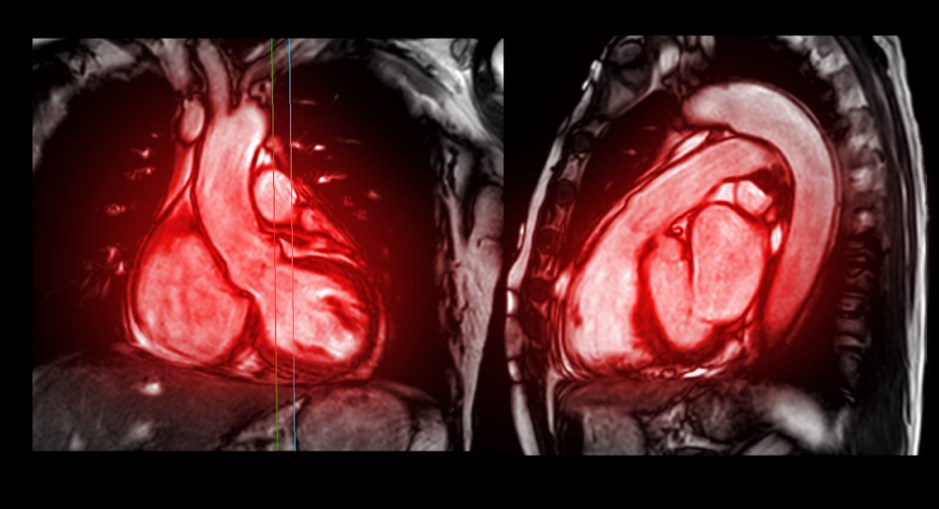
Research funded by the British Heart Foundation using novel cardiac imaging techniques was described in a recent article in Circulation. These techniques, such as quantitative perfusion cardiac magnetic resonance imaging and cardiac diffusion tensor imaging, can detect pre-clinical hypertrophic cardiomyopathy (HCM) before the development of any thickening in the heart.
With novel treatments like myosin modulators now available to patients, and with experimental treatments such as gene therapy now on the horizon, these new imaging techniques could allow doctors to intervene at a much earlier stage in order to potentially prevent disease.
You can read more about these imaging techniques here.