The January recall of 48 Medtronic CRT-D and ICDs has now been expanded to include 752 additional devices at lower risk than those involved in the January recall,. There is an issue that occurred during the manufacturing process of these devices which could result in an unexpected loss of device functionality. If you have one of these devices, you should contact your doctor to discuss next steps.
You can see the advisory here.
To look up your device by product name, model or serial number to see whether it is impacted, click here.
The box in the upper right corner labeled “Advisories For This Model” will tell you if there are any advisories for your device.
If you are affected, the search page would look like this:
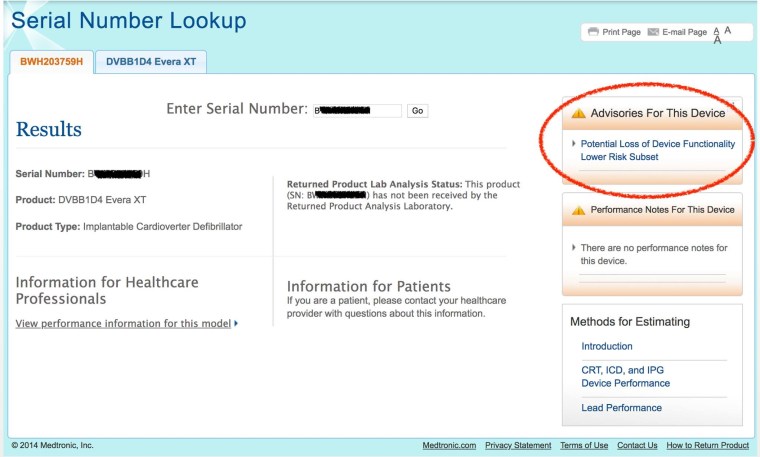
This example shows that currently there are no advisories for my model.
As always, you can call Medtronic Patient Services with any questions at: (800) 551-5544 (M – F, 8am – 5pm Central)

